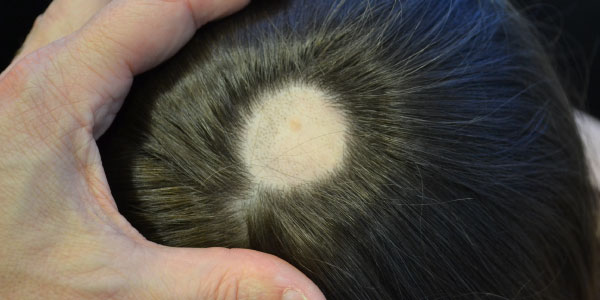
Individuals who have a diagnosis of severe alopecia areata (AA) may qualify for this clinical research trial and receive investigational oral medication or placebo.
- Age: ≥ 12 years and ≤ 65 years old
- 50% or more scalp hair loss
- Current AA episode less than 8 years
- No spontaneous scalp hair regrowth over the past 6 months
Patients will be asked to visit the clinic about 16 times over approximately 160 weeks. There is no cost to participate, and participants will be reimbursed for their time and travel.
For further information on the study, please follow the link:
Trial ID: NCT06012240
We are actively recruiting for this study. If you are interested and would like to participate in this clinical trial, please submit your details and our team will be in touch when it becomes available: expression of interest form
Accreditations & Memberships





















